|
Therefore, the user must decide which application is the most important for a specific task in order to determine the most compatible version of rechargeable batteries. Unfortunately, there are no batteries that are capable of encompassing all functions mentioned above. 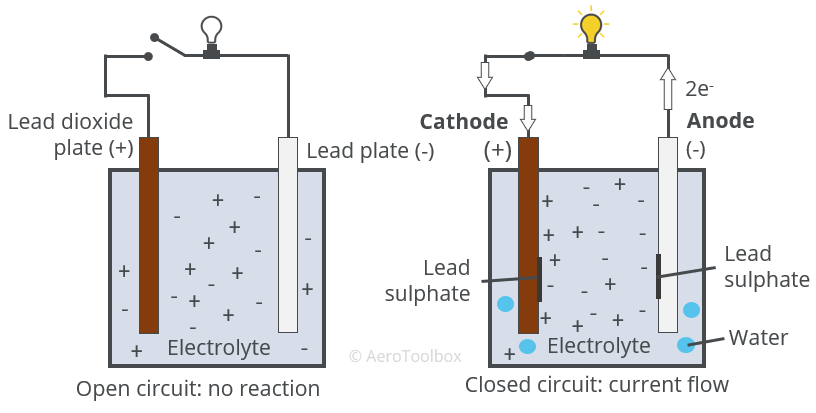
For long-term use (followed by discharging and charging), long storage time when not in use, remote activation, and use under harsh weather conditions are just a few obstacles of creating such secondary cells. This disequilibrium is the representation of the cell potential that, when allowed, could once again approach equilibrium through the transferring of the electrons.ĭifferent secondary batteries provide various functions. (Remember: Anode is the electrode on which oxidation takes place, i.e., loss of electrons and cathode is the electrode on which reduction takes place i.e., gain. This makes it seem like the positive charges are compiling on the other side of the cell which is not allowed to pass the separator. The Charger shown on the top of the diagram is pulling the negative charges toward the right side of the separator. The positive: PbO2 + 2 + HSO4 - e + 3 h + PbSO4 + 2 h2o. The figure below illustrates the mechanism of a charging secondary cell. The lead storage battery is made up of a group of lead-bismuth alloy grids filled with spongy metal lead, and the other group is made of lead-bismuth alloy grid filled with alumina. To simulate lead-acid battery (LAB) charging has never been an easy task due to the influences of: (1) secondary reactions that involve gas evolution and recombination and grid corrosion, (2. 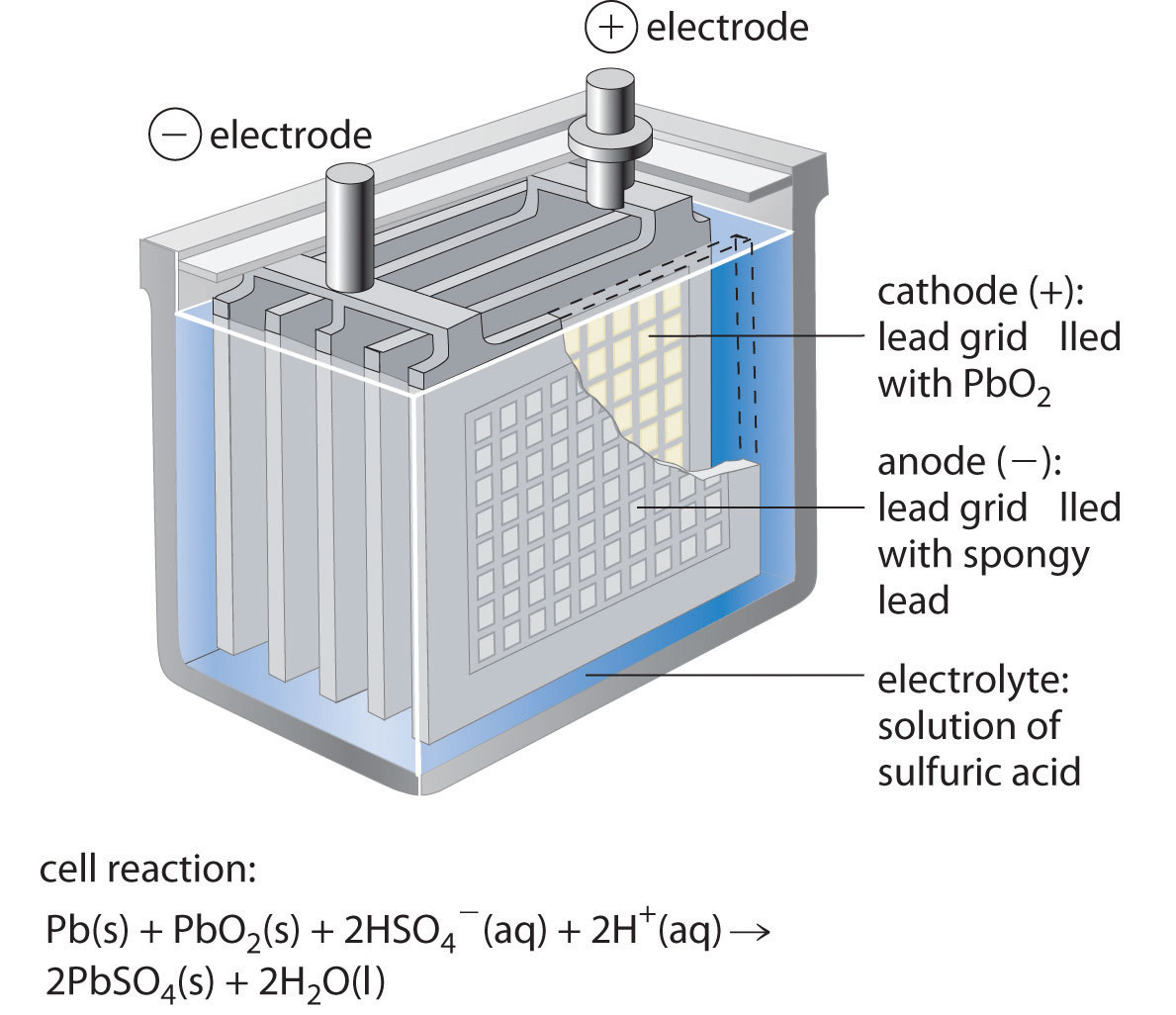 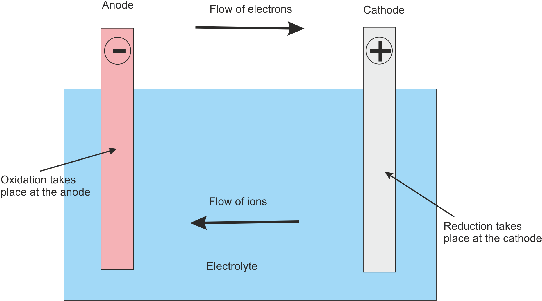
Secondary cells encompass the same mechanism as the primary cells with the only difference being that the Redox reaction of the secondary cell could be reversed with sufficient amount of energy placed into the equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
